Reactivity in Chemistry
Reaction Kinetics
RK3. Activation Barriers
Why do reactions take place at different rates? Why do some happen quickly, and others proceed very slowly? Why might the same reaction proceed at different rates under different conditions? There are a number of factors that influence reaction rates, but the first one that we will look at is the activation barrier.
An activation barrier is a sort of energetic hurdle that a reaction must get over. Some reactions have higher hurdles and some have lower hurdles. It's much easier to get over lower hurdles, so reactions with low activation barriers can proceed more quickly than ones with higher activation barriers.
A low activation barrier allows a reaction to happen quickly.
A high activation barrier makes a reaction go more slowly.
A reaction can be exergonic overall (it can give off energy), but it will generally still have an activation barrier at the beginning. Even if the compounds go down in energy by the end of the reaction, they will generally go up in energy before that happens.
Even if a reaction gives off energy overall, some energy must be put in at first to get the reaction started.
This situation is a little like investing in a business. A business generally requires some sort of financial investment to get started. If the business is successful, it will eventually make products and pay money back to the investors. If the business is unable to make back its initial investment, it may fail.
Reactions require some initial investment of energy. This energy may come from surrounding molecules or the environment in general. If the reaction is successful, it will proceed to make products and it will give energy back to its surroundings.
It always "costs" a molecule energy to enter into a reaction; it "borrows" that energy from its environment.
That initial investment of energy may be "paid back" as the reaction proceeds.
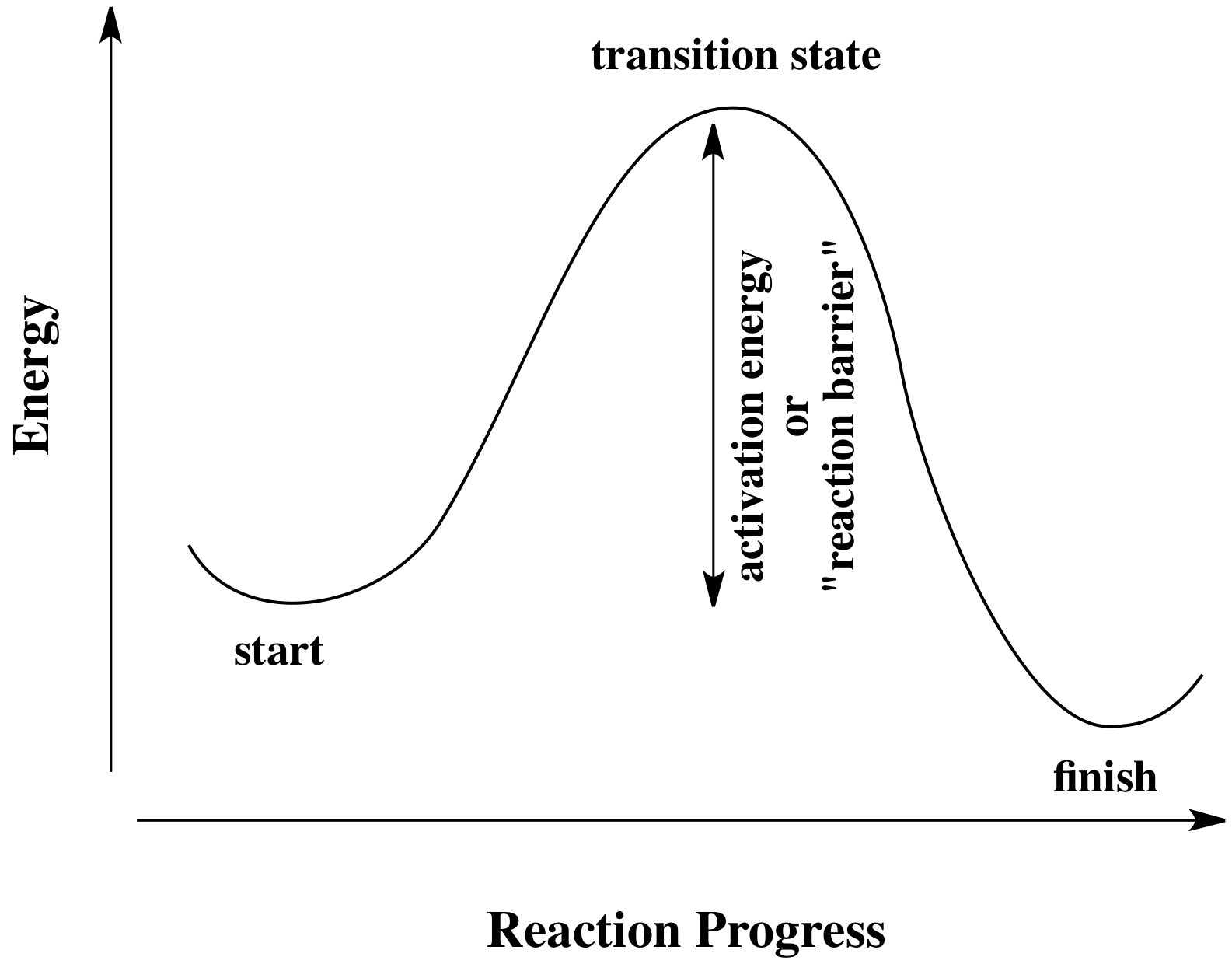
All reactions must overcome activation barriers in order to occur. The activation barrier is the sum of the energy that must be expended to get the reaction going. An activation barrier is often thought of, cartoonishly, as a hill the molecule has to climb over during the reaction. Once, there, it can just slide down the other side of the hill to become products. At the top of the hill, the molecule exists in what is called the "transition state". At the transition state, the structure is somewhere between its original form and the structure of the products.

Figure RK3.1. A reaction progress diagram showing the initial energy barrier in a reaction.
The type of diagram shown above is sometimes called a "reaction progress diagram". It shows energy changes in the system as a reaction proceeds. One or more activation barriers may occur along the reaction pathways, as various elementary steps occur in the reaction.
In order to see more concretely what terms like "reaction progress" and "transition state" mean, let's look at a real reaction. Suppose a nucleophile, such as an acetylide ion, donates its electrons to an electrophilic carbonyl. The π bond breaks and an alkoxide ion is formed.
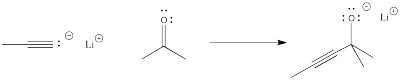
Figure RK3.2. A reaction of a nucleophile with an electrophile.
The reaction progess simply refers to how far the reaction has proceeded. Is it just starting out, is it almost finished, is it just halfway there? The transition state refers specifically to the highest energy point on the pathway from reactants to products. It refers to the structure at that point, and the energy associated with that structure.
In the following diagram, the term "reaction progress" has been replaced by an illustration that shows how far the reaction has proceed by that point in the energy curve that is above the reaction drawing. The structure in the square brackets is the transition state, coresponding to the highest point on the curve. The "double dagger" symbol (a little bit like a Patriarchal or Russian Orthodox cross, with two crosspieces on a vertical post) is the symbol that tells you that you are looking at a transition state structure.
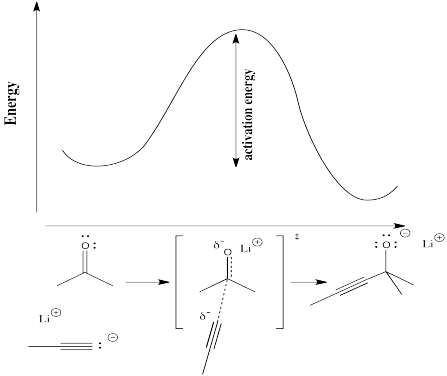
Figure RK3.3. A reaction barrier in a reaction, with "reaction progress" shown as structures along the reaction pathway.
The transition state doesn't refer to a regular chemical structure. It doesn't necessarily obey the rules of Lewis structures, because some new bonds have started to form and some old bonds have started to break; you can't really draw partial bonds in a Lewis structure.
Physically, the transition state structure is not something that can be isolated and stored in a bottle. Because it sits at the top of an energy curve, the transition state is motivated to turn into something else. No matter what direction it goes to change its structure, it will go to lower energy. Remember, things always proceed to lowest energy if possible. As soon as the transition state forms, it will either slide back into the original starting materials or slip forward into the final products.
The transition state is inherently a high-energy, unstable structure, with a very short lifetime. As soon as it comes into existence, it disappears again.
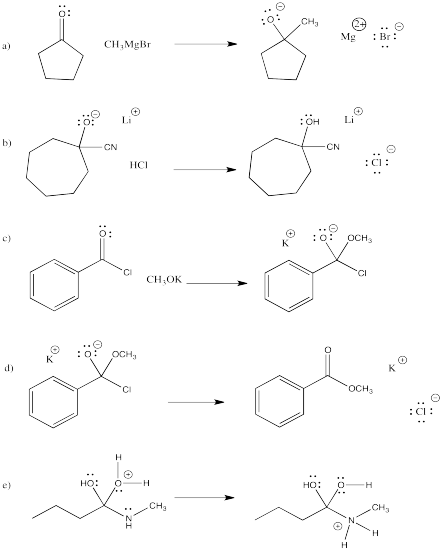
Problem RK3.1.
Draw what you think the transition state might look like for the following elementary reactions.
More commonly, reaction progress diagrams aren't drawn like the one above. Instead, structures of reactants, transition states and products are simply shown along the potential energy curve, as shown below.
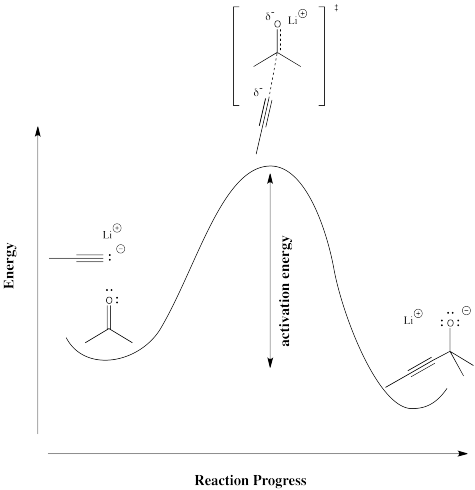
Figure RK3.4. Another way of drawing the reaction progress diagram.
Reactions don't always happen in one step. Sometimes there is an intermediate, or more than one. An intermediate differs from a transition state in that it has finite lifetime. Although it is not as stable as the reactants or the products, it is stable enough that it does not immediately decay. Going either forward to products or back to reactants is energetically uphill.
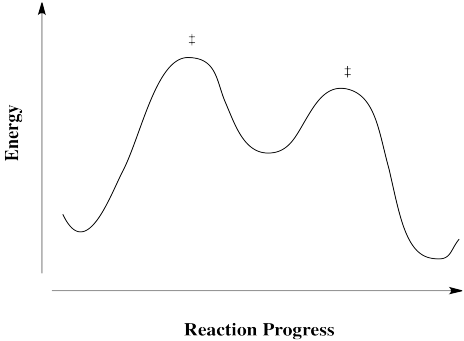
Figure RK3.5. A reaction progress diagram with an intermediate along the pathway.
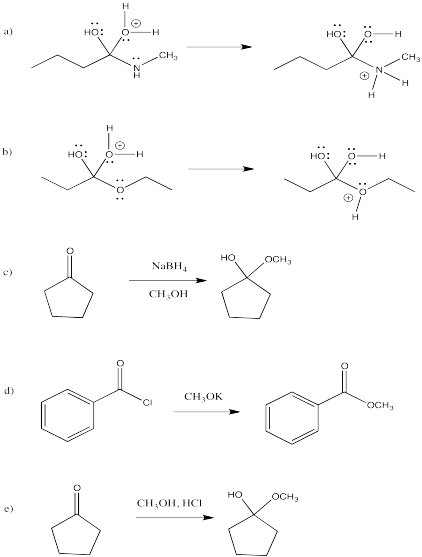
Problem RK3.2.
Draw reaction progress diagrams for the following reactions. Note that the reactions may be composed of more than one elementary step.
This site was written by Chris P. Schaller, Ph.D., College of Saint Benedict / Saint John's University (retired) with other authors as noted on individual pages. It is freely available for educational use.

Structure & Reactivity in Organic,
Biological and Inorganic Chemistry by
Chris Schaller
is licensed under a
Creative Commons Attribution-NonCommercial 3.0 Unported License.
Send corrections to cschaller@csbsju.edu
This material is based upon work supported by the National Science Foundation under Grant No. 1043566.
Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Navigation:
Back to Web Materials on Structure & Reactivity in Chemistry