Reactivity in Chemistry
Electrophilic Aromatic Substitution
AR1. Introduction to Electrophilic Aromatic Substitution
Aromatics, or arenes, are derivatives of benzene or other compounds with aromatic ring systems. That is, they are cyclic, planar, fully conjugated and have an odd number of π-electron pairs. Like alkenes, aromatics have π-electrons that are loosely held and are easily attracted to electrophiles. However, aromatics don't undergo the typical reactions of alkenes.
For example, bromine adds very quickly to the double bond of an alkene. If you take a (generally colourless) alkene and dissolve it in a solvent, then add a solution of bromine drop by drop to the solution of alkene, you can see the reaction happen before your eyes. Br2 is a deep red-brown. When a drop of the bromine solution hits the alkene solution, the red-brown colour disappears almost instantly as the Br2 is consumed.
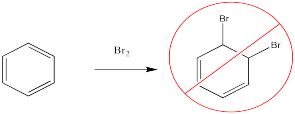
In contrast, bromine will not add across the double bond of benzene. If you do the same experiment with a solution of bromine and a solution of benzene, the red-brown colour will persist.

Figure 1.1. The lack of electrophilic addition across double bonds of benzene.
To be clear, Br2 can react with benzene, but it won't happen as simply as adding one to the other like it will with an alkene. Instead, under the right conditions, a bromine atom replaces one of the hydrogen atoms on the benzene. This won't normally happen just by adding bromine to benzene. The reaction of benzene with bromine is extremely slow. The reaction is greatly accelerated, however, in the presence of Lewis acids, such as ferric chloride.
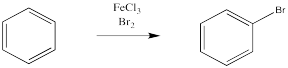
Figure 1.2. The use of a Lewis acid catalyst in the bromination of benzene.
A similar reaction happens with chlorine. Cl2 will not react directly with benzene like it will with an alkene. On the other hand, if a benzene solution is treated with chlorine gas and a metal catalyst, a chlorine atom from the Cl2 can replace a hydrogen atom on benzene. However, the same thing doesn't work as smoothly with the other halogens, iodine and fluorine (although other methods can be used to make fluorobenzene and iodobenzene).
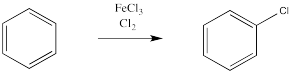
Figure 1.3. The use of a Lewis acid catalyst in the chlorination of benzene.
The reactions of chlorine and bromine with benzene and other aromatics can be catalysed by a variety of Lewis acidic metal catalysts. These Lewis acid catalysts can activate these halogens, making them much more able to attract electrons from the benzene.
Halogens are not the only electrophiles that can be activated by Lewis acids. Lewis acids can also induce the reactions of benzene with alkyl halides and acyl halides. We don't normally think of alkyl halides and acyl halides as electrophiles for alkene addition. We do think of them as electrophiles under other circumstances. When we think of an SN2 reaction, we are thinking of an alkyl halide electrophile. When we look at substitution of a nucleophile at a carboxyloid, the simplest examples use acid chloride electrophiles. Alkenes are a different matter. They aren't appropriate nucleophiles for alkyl halides. However, under some circumstances, alkyl halides can be activated, leading to cationic intermediates that can react with alkenes; some alkene polymerizations are conducted this way. A similar strategy has to be employed in order to get the even less reactive benzene to react with an alkyl electrophile.
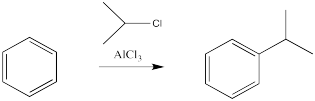
Figure 1.4. The use of a Lewis acid catalyst in the alkylation of benzene.
There are some limitations on what kind of groups can be added in this way. The carbon attached to the halide should be tetrahedral. Typically, it is much easier to add secondary or tertiary alkyls than primary ones. That is, the carbon attached to the halogen had best be attached to two or three other carbons as well. Methyls are very, very difficult to add in this way.
There is an important exception. The carbon attached to the halogen need not be tetrahedral, provided it is a carbonyl carbon. That reaction is called an acylation.
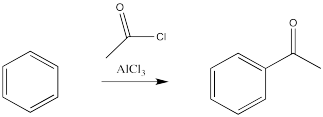
Figure 1.5. The use of a Lewis acid catalyst in the acylation of benzene.
In these cases, it is the alkyl or acyl, rather than the halogen, that replaces a hydrogen atom on the benzene. Remember, benzene is most likely acting as a nucleophile in this reaction, even though it is following a different pathway than an alkene would. It is reacting with the most electrophilic part of the alkyl halide or acyl halide.
Aromatics have a limited repertoire of electrophiles with which they commonly undergo reaction. In addition to these Lewis acid-catalysed reactions, there are also reactions strong acidic media, such as a mixture of nitric and sulfuric acid.
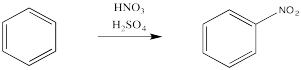
Figure 1.6. The use of a sulfuric acid catalyst in the nitration of benzene.
Another acidic medium, referred to as "fuming sulfuric acid", is really a mixture of sulfuric acid and sulfur trioxide.
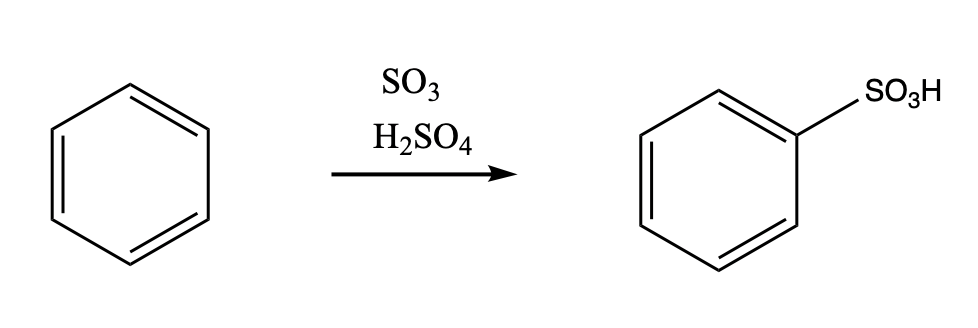
Figure 1.7. The use of a Lewis acidic sulfur trioxide in the bromination of benzene.
Just as with the acid-catalysed reactions, the nitro group and the sulfonate group just replace a hydrogen atom on the benzene ring. The overall reaction involves bond formation between a benzene carbon and the electrophile, and bond cleavage between the same carbon and a proton.
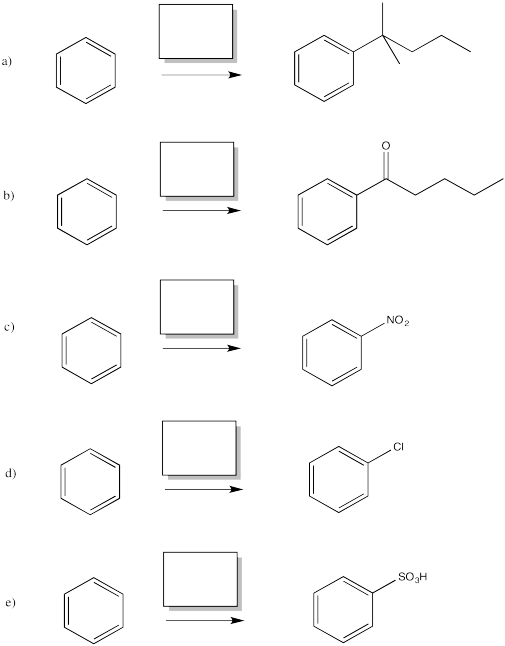
Problem AR1.1.
Fill in the missing reagents in the following reactions.
This site was written by Chris P. Schaller, Ph.D., College of Saint Benedict / Saint John's University (retired) with other authors as noted. It is freely available for educational use.

Structure & Reactivity in Organic, Biological and Inorganic Chemistry by Chris Schaller is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported License.
Send corrections to cschaller@csbsju.edu
This material is based upon work supported by the National Science Foundation under Grant No. 1043566.
Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Navigation: