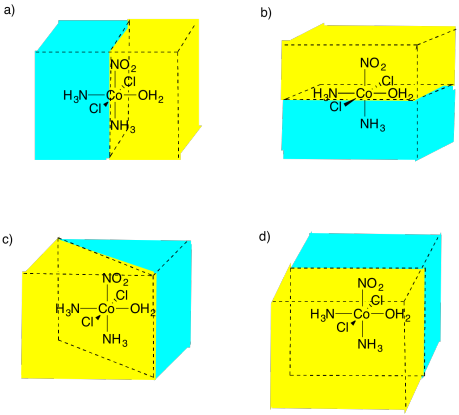
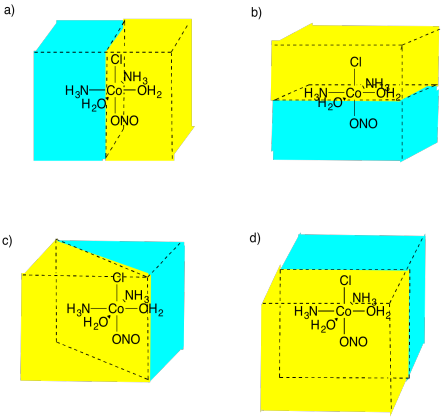
SC3. Enantiomers
An atom with four groups attached to it can also adopt a tetrahedral geometry. This geometry often occurs when the central atom is a little smaller. A tetrahedral geometry allows neighbouring groups to get a little farther from each other. The following model of methane shows a pretty simple example. The grey ball is a carbon atom and the four white balls are hydrogen atoms.

Figure SC3.1. A ball-and-stick model of a simple tetrahedral compound: methane, CH4.
Go to Animation SC3.1. A three-dimensional model of methane, CH4.Unlike square planar compounds, simple tetrahedral compounds do not have the same kind of cis- and trans- isomers. That is, two groups can't be placed on a tetrahedron so that they are opposite each other or beside each other. The relationship between any two groups on a tetrahedron is the same as the relationship between any other two groups on a tetrahedron.
Dichlorodimethylsilane is a compound that can be used to make silicone polymers. Like platin, it has two each of two groups attached to the central atom. However, the central tom is tetrahedral. There is only one way to arrange these four groups.
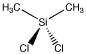
Figure SC3.1. A tetrahedral atom with two different types of groups attached, (CH3)2SiCl2.
This compound is called dichlorodimethylsilane. There are two chlorines and two methyl groups attached to the central silicon atom. On the previous page, we saw a pretty similar pair of compounds, cis-platin and trans-platin. Those compounds were square planar at the central platinum atom. In a square planar situation, two groups could either be attached to the central atom right beside each other (they would be cis to each other) or across from each other (they would be trans to each other). In a tetrahedral case, these differences are not possible because two group are always exactly the same distance away from each other.
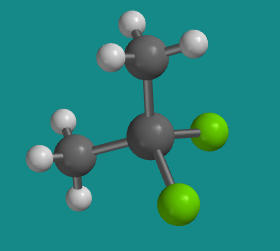
Figure SC3.2. A ball-and-stick model of dichlorodimethylsilane.
Go to Animation SC3.1. A three-dimensional model of dichlorodimethylsilane.
However, if four different groups are attached to a tetrahedral atom, the four groups can be arranged in two possible ways. The two compounds that result are mirror images of each other. These two isomers are called enantiomers.
For example, suppose that tetrahedral silicon atom had four different groups attached to it: a hydrogen atom, a chlorine atom, a methyl group (that's a carbon with three hydrogens), and a hydroxy group (that's an oxygen with one hydrogen). There would be two slightly different ways to organize those four groups in space. In the picture below, hydrogen is white, chlorine is green, and oxygen is red, whereas silicon and carbon are both grey.
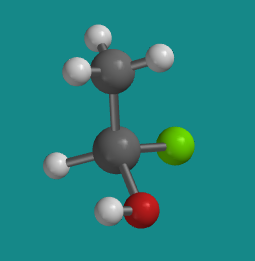
Figure SC3.3. A ball-and-stick model of R-chlorohydroxymethylsilane, one of two stereoisomers.
Go to Animation SC3.2. A three-dimensional model of R-chlorohydroxymethylsilane.
Problem SC3.1.
Turn the model so that the hydrogen attached to the silicon is hidden behind the molecule. In what direction would you travel if you traced from the chlorine to the oxygen to the carbon: clockwise or counter-clockwise?
This next picture shows a very similar molecule. The same groups are attached to the central silicon atom, but the are arranged in a slightly different way.
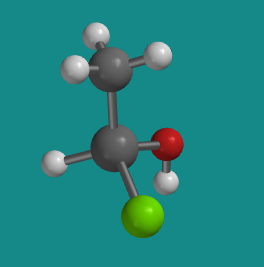
Problem SC3.2.
Turn the model so that the hydrogen attached to the silicon is hidden behind the molecule. In what direction would you travel if you traced from the chlorine to the oxygen to the carbon: clockwise or counter-clockwise?
Full disclosure: the molecule we are looking at isn't really a stable compound that you could pour out of a bottle from the chemistry stockroom. That's because one molecule would react with another one just like it, and then another one, and so on to form a long chain called a silicone polymer. That's the kind of material found in things like silly putty and silicone caulking.
Problem SC3.3.
Let's try working without the three-dimensional model. The following ball-and-stick picture shows a silicon attached to a bromine (red), a chlorine (green), a fluorine (yellow) and a hydrogen. If you were looking at the molecule with the hydrogen hiddent behind it, in what direction would you travel if you traced from the bromine to the chlorine to the fluorine: clockwise or counter-clockwise?
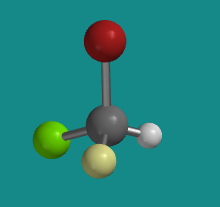
Problem SC3.4.
If you were looking at the molecule with the hydrogen hiddent behind it, in what direction would you travel if you traced from the bromine to the chlorine to the fluorine: clockwise or counter-clockwise?
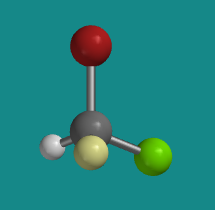
There don't have to be four different atoms attached to the central atom to get two stereoisomers. There just have to be four different groups. Even if all four atoms attached to the central one are the same, if they have different things attached to them, then two three-dimensional arrangements are possible. The following compounds illustrate a more subtle example like that. This compound, by the way, is stable and can be isolated and stored; it's a real compound.
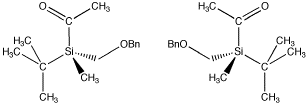
Figure SC3.5. A pair of enantiomers. The (-) enantiomer is on the left and the (+) enantiomer is on the right. Note that the tetrahedral silicon atom has four different groups attached.
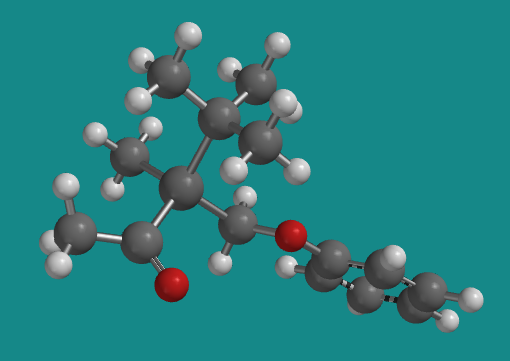
Figure SC3.6. Another view of the (-) enantiomer; it's in a different orientation from the wedge/dash structure above.
Go to Animation SC2.1. A three-dimensional model of the (-) enantiomer.
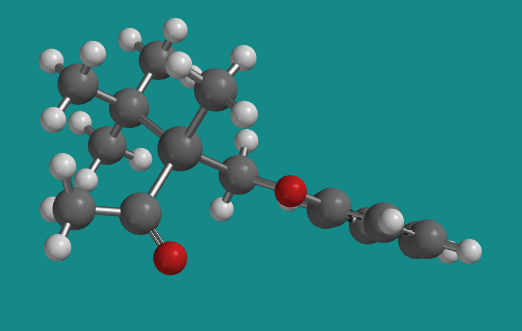
Figure SC3.7. Another view of the (+) enantiomer; it's in a different orientation from the wedge/dash structure above.
Go to Animation SC2.1. A three-dimensional model of the (+) enantiomer.
- Enantiomers are pairs of compounds with exactly the same connectivity but opposite three-dimensional shapes.
- Enantiomers are not the same as each other; one enantiomer cannot be superimposed on the other.
- Enantiomers are mirror images of each other.
Two compounds with the exact same connectivity, that are mirror images of each other but that are not identical to each other are called enantiomers. The more common definition of an enantiomer is that it is not superimposable on its mirror image. It can be distinguished easily from its mirror image, just as a right hand can easily be identified and distinguished from a left hand.
- Compounds that occur in these pairs are called "chiral".
- "Chiral" comes from the Greek word for "hand".
It can be shown using group theory, the mathematics of symmetry, that an enantiomer may also be defined as a molecule that does not contain a mirror plane, meaning it cannot be divided into two identical and opposite halves.
- Enantiomers contain no mirror planes.
- Enantiomers do not contain two equal and opposite halves.
Unlike cis- and trans-isomers, two enantiomers have the same physical properties. they have the same melting point, the same solubility, and so on. Two compounds that are almost identical, but mirror images of each other, have exactly the same kinds of intermolecular attraction, so it may not be a surprise that their physical properties are identical.
- Enantiomers are another example of a type of stereoisomers.
- Two enantiomers have identical physical properties, except for optical rotation.
Optical rotation involves the interaction of plane-polarized light with a material. If a material is not symmetric, the light that passes through it will be rotated. That means if the waves making up the light are oscillating in one direction as they enter the material, they will have tilted slightly to oscillate in another direction when they emerge from the material. We will look at this phenomenon later.
- Two enantiomers have an equal but opposite rotational effect on plane-polarized light.
- (+) enantiomers rotate light in a clockwise direction.
- (-) enantiomers rotate light in a counterclockwise direction.
For example, in the chiral silicon compound shown above, the (+) enantiomer rotates plane-polarized light in a clockwise direction. It has a "standard optical rotation" of [a] = +12 ( +/-2)o. The (-) enantiomer rotates plane-polarized light in a counterclockwise direction. It has a "standard optical rotation" of [a] = -9.9 ( +/-2)o.
Problem SC3.5.
A certain compound exists in two forms; enantiomer A and enantiomer B. Enantiomer A has a molecular weight of 126 g/mol, a density of 0.995 g/mL, an optical rotation of [a] = 26o, a melting point of 65 oC, a boiling point of 225 oC, and an odour of citrus fruit. What can you say about the corresponding properties of enantiomer B?
Problem SC3.6.
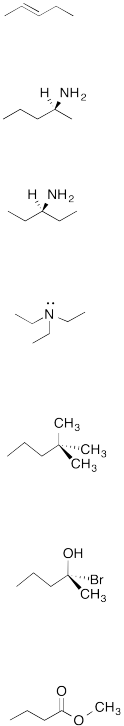
Are these molecules chiral?
Draw the mirror image of each compound and determine if it is superimposable.