Monomers & Polymers
MP3. Olefins
Polyolefins are made from "olefins", which you may know as alkenes. Olefin is an older term for an alkene that is still commonly used in industry.
These compounds make up a significant fraction of commercially-used polymers today. If you think of the common recyclable plastics, polyethylene (#2 and #4, depending on how the material is made), poly(vinyl chloride) (#3), polypropylene (#5) and polystyrene (#6) are all examples of polyolefins.
Polyolefins have been known for some time, although it took about a century from the time that they were first documented until they were recognized as polymers. German chemists in the mid-1800's were aware that certain tree resins gave rise to hard materials over time, and these materials included polystyrene.

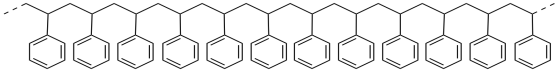
If you compare the repeating structure of polystyrene to the structure of styrene, you can imagine that polystyrene is made from a series of styrene molecules that have been strung together. Essentially, the double bond of styrene has moved to attach to the next molecule, and the double bond there has done the same thing, and so on.

Polyolefins are formed in the same way from a wide variety of alkenes, leading to an array of different materials with properties that are suited to unique applications. At the simplest level, ethene molecules can be attached to each other to form polyethylene.
![]()
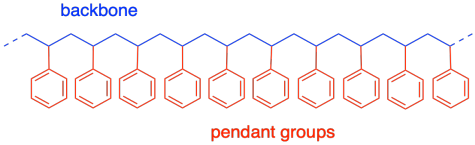
Most polyolefin structures have something in common with polyethylene. In general, a similar carbon backbone forms the main chain of this class of polymers. Polymers such as polystyrene also contain that backbone, but they have pendant groups ("hanging" groups) along that backbone. In the monomer, the pendant group was attached to one end of the alkene double bond.
Note that, like ring-opening polymerization, olefin polymerization depends on molecules reacting with other molecules that are just like them. That isn't likely to happen; they will need something to come along and induce reaction between them. Olefin polymerization is thus another case in which monomers are ted together through a chain reaction. The reaction will require an initiator to get things started. That initiator is likely to become an end group, hanging from one end of the polymer chain or the other.
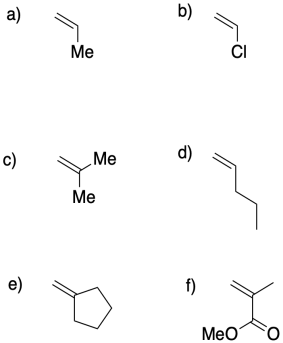
Problem MP3.1.
Draw structures of the polymers that are made from each of the following monomers.
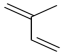
Problem MP3.2.
If you are familiar with the concepts of regiochemistry, stereoisomers and conjugate addition, then you may anticipate that there are several different polymers that could possibly result from the monomer, isoprene, below. Show these possibilities.
This site is written and maintained by Chris P. Schaller, Ph.D., College of Saint Benedict / Saint John's University (with contributions from other authors as noted). It is freely available for educational use.

Structure & Reactivity in Organic, Biological and Inorganic Chemistry by Chris Schaller is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported License.
Send corrections to cschaller@csbsju.edu
Support for this project was provided by the Opens Textbooks Pilot Program of the U.S. Department of Education through a collaboration with the Libre Texts project at University of California, Davis.
Navigation:
Back to Structure & Reactivity